5 Investigations
Pulmonary Function Lab
The Pulmonary Function Laboratory at Monash Health offers the whole breadth of pulmonary function tests:
Spirometry
Diffusion capacity measurement
Body plethysmography (lung volume measurement)
Oxygen assessments
Altitude simulation tests
Cardiopulmonary exercise testing (CPET)
The lab is also your go to address for organising oxygen equipment to be delivered to family’s homes.
Keeping track of things
Particularly for CF patients lung function results are an important marker of overall health and a key factor to direct treatment. Try to keep track of current, recent and previous (6 months) results of your patients. Your consultant is likely to enquire about these during ward rounds. You can also print off graphical representations of a patient’s key parameters over time (such as FEV1, FVC, weight etc.) via Respiro, the lung function database software available on the G-drive. Visual representation of data, if done right, is much better digested by our brains than a row of plain numbers. In addition, time-series of data gives a better indication of what is happening with your patient than a single data point.
Consider a single reading of an oxygen saturation of 94% in a patient you’re looking after. That’s no too bad - certainly nothing to worry about for the moment, you’d think. So let’s add a bit of clinical context.
You’re looking after a 4 week old baby corrected age, born at 28 weeks. She’s only been home for three weeks and presented to MCH ED a day ago with wheeze and crackles, in keeping with a diagnosis of early bronchiolitis. She also wasn’t feedings as well and pulse oximetry showed her to be hypoxaemic. She has since been doing well in subnasal O2 of 0.25 L/min, with her oxygen saturations reliably sitting above 95%.
Given the history of this baby, you’ll probably feel your Spider Sense[^6] going off by now. This is a vulnerable baby for a number of reasons, including extreme prematurity, early requirement of supplemental oxygen and - given the timeline of being early in this illness - a high likelihood for further deterioration. The fact that the oxygen flow that previously led to complete resolution of hypoxaemia is no longer enough should act as a strong trigger to suspect that things are currently going in the wrong direction for this baby.
Think about what your next steps would be to manage this baby (in addition to notifying your ATR).
Access to the respiratory database is organised by Paul Finlay in the lab - he’ll be happy to arrange access for you.
Oximetry has long become the 5th vital sign, standing proudly in a line with temperature, blood pressure, pulse and respiratory rate (though still competing with pain for this position).
Like with so many parameters there is added value in evaluating oxygen saturation and its evolution over time (“trend”). Recorded oximetry is often used in overnight recordings. Added pressures come into play in the sleeping child. Respiratory rate and tidal volume are reduced during sleep, potentially unmasking underlying respiratory insufficiency. Nocturnal changes in muscle tone can give rise to obstructive sleep apnoea which might be diagnosed using oximetry. Identifying multiple clusters of oxygen desaturation to levels below 90% is consistent with obstructive sleep apnoea in a patient whose history and clinical examination also fit the diagnosis. Unfortunately, the sensitivity of this test is only 50% when compared to a sleep study so a negative overnight oximetry is unable to rule out this condition.
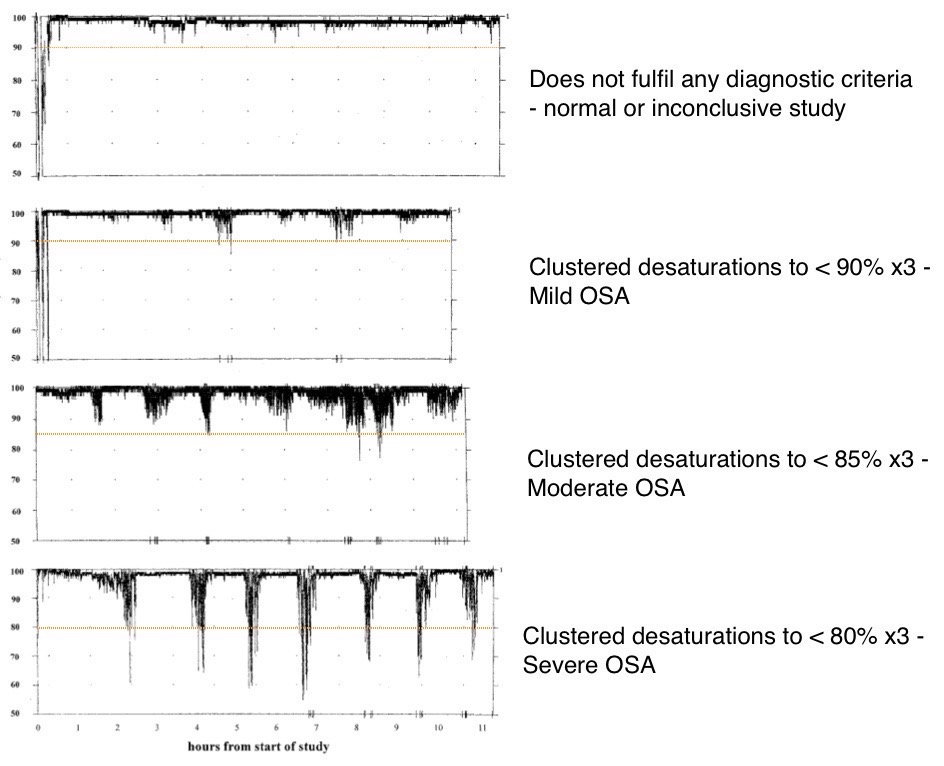
Organising an oximetry
In order to interpret overnight oximetry you will need an recording oximeter whose data can be downloaded into a computer for analysis. The oximeters on our wards are not equipped with this functionality and you will require one of the specialised recording oximeters from the Sleep Unit.
There’s more: not all oximeters are built equal. The signal quality that oximeters return is greatly impaired by subject movement. Manufacturers use proprietary algorithms to reduce the impairment in signal-quality arising from this. Despite what the oximeters make it look like, you do not actually see a live SaO2-reading on the display. Rather, oximeters display a mean of samples over a time-period (averaging time) so that the displayed numbers don’t jump around so much during periods of difficult signal acquisition. In many oximeters this averaging time is set to a default of 12 seconds. This is a useful setting to evaluate the trend of a patient presenting with an acute problem of cardiac output or oxygenation.
When diagnosing obstructive sleep apnoea we have more specific needs: we are looking for potentially very short-lived oxygen desaturations caused by airway obstruction. An ideal device would obtain a “beat-to-beat” analysis of arterial oxygen saturations. No oximeter currently on the market offers an averaging time this short.
We have found that Masimo brand oximeters work best for our purposes. Their artefact suppressing technology is effective while preserving valuable signal data. And: the averaging-time can be set as low as two seconds.
In short: not every oximeter will do and the choice of machine might differ based on the clinical question you want answered. Keep these limitations in mind when presented with oximetries performed outside of our Sleep Unit.
Inpatient oximetry
Monash Children’s Hospital paediatric wards
When organising an overnight oximetry for an inpatient get in touch with the Children’s Sleep Centre:
• Nina Lyons, x23593
• Bella Cortes, x23592
• Rebecca Mihai, x23589
• Nicole Verginis, x23589
Most of the Children’s Sleep Centre’s recording oximeters are booked out in advance for outpatient studies. Planning ahead and letting the unit know early is key to securing a device. Occasionally, a unit might be available on the same day but this will be a rare event. You will need to fill out an oximetry request form (available at the Sleep Centre or from mcsu.org.au → Referrals). Discuss the request with the ATR or on-call consultant and document specifics on the request form.
 |
Discharge process for infants with chronic neonatal lung disease on home oxygen therapy |
Neonatal Intensive Care Unit (NICU)
For babies in the NICU you should liaise with the Monash Newborn registrar responsible. Newborn Services are structured by campus (Monash, Dandenong, Casey). With the Monash unit being so big (65 beds), it is further structured into NICU and Special Care Nursery services, each sub-divided into teams.
Newborn services at the Monash Campus have their own recording oximeters and can usually organise oximetry to happen the very same night. They will also print off the recording for you.
This is not usually the case for babies at the Casey and Dandenong campuses. In order to obtain an overnight oximetry for babies at these campuses, the Casey and Dandenong services will usually need to request an oximeter from Monash Children’s @ Home.
What to request
As always, be specific in how you want the study to be performed:
Position of baby - prone or supine?
Room air or supplemental oxygen? If you would like supplemental O2, specify the flow required.
At times it may be helpful to “split” the night. You might order an oxygen flow of 0.125 l/min from 7pm to midnight and 0.25 l/min from midnight to 7am. This might save the need for another night’s recording.
It is rarely helpful to record overnight oximetry in oxygen flows < 0.125 l/min as the oxygen equipment used in the family’s home can not reliably deliver flows less than 0.125 l/min. That said, the flow meters in the NICU cannot be set to 0.125 l/min and we will use a satisfactory oximetry in 0.1 l/min as an indicator for a flow of 0.125 l/min at home.
After the oximetry
If you used an oximeter from the Children’s Sleep Centre, please collect it first thing after morning handover and return it to the unit. It will be downloaded and cleaned so that it can go back into service in the early afternoon. The unit will print off the recording for you and have it ready for you to collect later in the day. Should you require the recording urgently discuss with one of the scientists but be mindful of the fact that they may have other things to do first.
Make sure the ATR/consultant know that the study took place and inform them about any problem that occurred during the recording. Once you have the study printout discuss it with the ATR or on call consultant. An easy way of ensuring the consultant has a copy of the report is to scan and email it. The sleep unit can do this for you, if you ask them.
Oximetries and medical records
Recorded oximetries are often a central part of deciding on the requirement for supplemental oxygen or respiratory support for a patient.
Sleep lab staff will upload all oximetries and sleep studies to SMR – but not into our EMR. For inpatients having oximetry or sleep study enter a note into the EMR to direct other teams caring for this patient to look up results in SMR.
Oximetry requests from non-network hospitals
From time to time, the ATR will receive requests to interpret oximetries obtained at and sent in from non-network hospitals. Make sure that the oximetry fulfils some basic criteria:
Averaging time of oximeter was set to lowest setting (ideally 2s).
A bedside chart accompanies the recording that details times when the baby was crying, nappy was changed, position of baby in cot, O2 flow used (if any), etc. This greatly improves interpretation of the data.
Referrers should download a request form from https://bit.ly/mchoximetry
Please ensure the oximetry and relevant attachments are forwarded to the sleep centre. Once reported, the ATR will scan the report to Monash SMR and forward the results to the requesting consultant.
In selected patients, the we might decide to use a device to monitor an infant for the occurrence of central apnoeas. You know these as apnoea monitors or “Graseby-monitors”, named after a manufacturer of these devices. Unfortunately, they are now out of production. As there is no replacement on the market that meets our requirements, we still have a number of these older monitors in use.
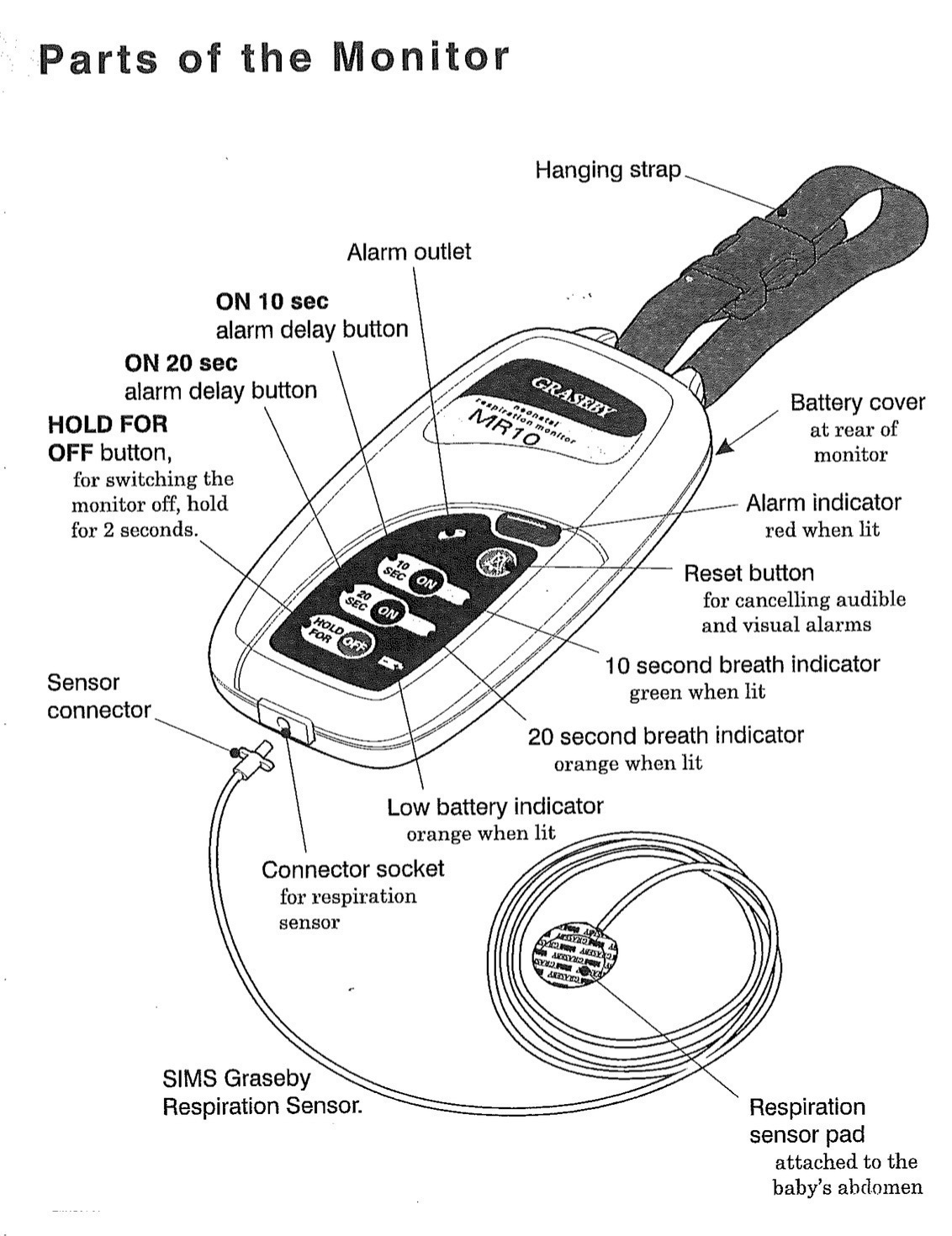
A small sensor with an air-cushion is attached to the baby’s abdomen with adhesive tape. Abdominal excursions during the baby’s breathing cause small pressure fluctuations in the sensor’s air cushion. The apnoea-monitor detects these pressure fluctuations and expects at least one such fluctuation every 10 or 20 seconds (depending on the chosen setting). If there is no fluctuation within the set time-period the monitor will take this as cessation of breathing and alarm.
Which baby needs an apnoea monitor?
The users of apnoea alarms within Monash Children’s mostly fall into one of two groups:
Infants following life threatening events
Infants with documented central apnoeas (respiratory drive disorders)
There will be the occasional baby outside of these groups we will consider an apnoea monitor for (e.g. sibling of infant with SUDO (Sudden Unexplained Death in Infancy) or a young infant required to be slept prone. The consultant in charge will decide on the appropriateness of using an apnoea monitor for a given child.
Apnoea monitors are not useful in babies presenting with obstructive apnoeas. These babies will show ongoing respiratory effort (thus not triggering the monitor) but markedly impaired ventilation and potentially significant hypoxaemia.
❖
How to arrange for an apnoea monitor
An apnoea-monitor needs to be requested by a Respiratory or Sleep consultant. Requests from other units need to be discussed with the Respiratory consultant on call before you can progress along this list.
Obtain approval for monitor loan from Drs Davey (x23586) or Nixon (x23587) who oversee the NIV/monitoring-program.
Ensure monitor availability with the administrative assistants (x23592/23593) in the paediatric sleep lab. The lab will require the following information:
Name of requesting consultant
Details of patient and parents - ideally patient sticker
Whether consultant will pick up the monitor or to send monitor to consultant’s office
Organise resuscitation training for all relevant care-givers. For ward patients, this should be facilitated through ward staff. Outpatients can be directed to St. John’s Ambulance Service[^7] (Phone 8588 8590).
Inform the consultant about the outcome. The requesting consultant needs to personally handover the monitor to the family to ensure the family know how to operate it correctly. This can be done in clinic or on the ward. The consultant may ask you to organise a time.
Things to know
The Sleep Centre only has a limited number of apnoea-monitors available. Accordingly, we need to make sure use of a monitor is indeed required (→approval from Drs Davey or Nixon).
There is no Medicare reimbursement to the unit for the cost of maintaining these devices. In order for us to continue offering this service, parents are required to rent these monitors. Monthly cost to the family is $50. There is also an upfront $100 deposit payable (reimbursed following return of the monitor). Sensors cost $12 each (they age with frequent re-taping and need regular replacing). Total cost for the first month will be $186 (Deposit $100 + 1st month’s rent $50 + 3 sensors $36).
Apnoea monitors can help indicate a baby that requires intervention or review by the caregiver. They are in no way a guarantee that an infant will not come to harm. Be mindful of your wording of what these monitors can achieve.
The unit gives out a family booklet on the use of apnoea-monitors and which also contains a resuscitation algorithm. Have a browse through - it is always helpful to know which information families receive so you can answer questions if required.
Alternatively, a monitor can be rented directly by the family from Smiths Medical in Sydney for comparable cost. www.smiths-medical.com, ☏ (02) 9634 9200.
Background
We frequently discharge infants who still require supplemental oxygen. Supplying families with domiciliary oxygen is a standard procedure nowadays but is actually one of the more recent approaches to out of hospital care for neonates and infants.
The original use of home oxygen was in adults with COPD following studies in the 1960s showing reduced mortality in this group, when treated with supplemental oxygen. In 1976, Pinney and Cotton[^8] published a paper on the home use of oxygen for children with bronchopulmonary dysplasia, facilitating earlier discharge from hospital. Children’s Hospitals all over the world followed the example, building their own home oxygen programs. Today, the requirement for supplemental oxygen is no longer a reason for a child to be stuck in hospital.
The majority of home oxygen requests flow through our department. The most common indication for the use of home oxygen is in preterm children with oxygen dependant chronic neonatal lung disease (CLD). Definitions for CLD vary but a good working definition is ‘the requirement for supplemental oxygen beyond 36 weeks post-conceptional age’.
Referral process
Children will mostly be referred from Monash NICU or network SCNs (Dandenong Hospital, Casey Hospital). Referrals may also come from other specialty teams. In general, the pathway to setting up a child on home oxygen is as follows:
Introduction
Monash Children’s Sleep Centre is the biggest paediatric sleep lab in the state. We conduct four diagnostic and an additional research polysomnography (PSG) five nights a week when not actually hampered by pandemic limitations. In addition, the centre offers Multiple Sleep Latency Tests (MSLTs) - a labour-intensive diagnostic test used in the workup for some conditions presenting with excessive daytime sleepiness (EDS), such as narcolepsy.
A portable sleep study unit is available for patients who are too unstable to be studied in the sleep lab. Please discuss with Drs Davey / Nixon regarding staffing and availability.
PSG captures neurological, cardiovascular and respiratory parameters during sleep that can be assessed by non-invasive means. Standard recordings include:
Limited channel electroencephalogram (EEG)
Electrooculogram (EOG)
Electromyogram (EMG, chin and leg)
Electrocardiogram (ECG)
Chest and abdominal excursion using respiratory inductance plethysmographic (RIP) belts
Pulse oximetry
Oro-nasal airflow via pressure transducer and thermistor
Capnography via transcutaneous and end-expiratory CO2
Continuous audio and video recording
Automatic position monitor, to record body position (B/L/R/F)
The setup-up process is quite involved. Experienced paediatric sleep technicians take about 45 minutes to place the sensors on an older child. In younger children or those who feel more anxious, have more difficulty adjusting to new environments or unfamiliar sensations to their skin this process can take much longer. The unit is always happy to show around a family prior to a study to ease the feeling of “the big unknown”. Call the unit’s administrative assistants to arrange a suitable time.
 Some more details you should know about
Some more details you should know about
Some more details you should know about
Families are asked to arrive at the lab at 7.30 pm. Studies run until 6 am when the families are woken.
Each child has their own study room. A parent stays with them overnight in a separate bed.
Night staff consists of a mixture of sleep scientists and nursing staff. Complex or potentially medically unstable patients booked for PSG require prior discussion with Drs Nixon or Davey to allocate appropriate staff.
What is assessed
When you look at the parameters recorded during a PSG you can somewhat delineate the role PSG plays in the diagnostic process of sleep disorders.
EEG in conjunction with OMG is used to identify the child’s sleep stage. Sleep is divided into two broad categories: REM-sleep (active sleep in infants) and NREM-sleep (quiet sleep in infants), which is further sub-divided into NREM-1, NREM-2 and NREM-3 based on EEG characteristics).
REM (rapid eye movements) sleep is a sleep phase characterised by rapid eye-movements, low-muscle tone and often vivid dreams. It has essential homeostatic functions and is often preserved even in severely sleep deprived individuals. Newborns spend around 50% of sleep in REM. Once the brain is fully matured, 20% of total sleep time is spent in REM.
NREM-1 sleep marks the transition between wakefulness and sleep. Brain waves transition from alpha-waves (8-13 Hz) to theta waves (4-7 Hz). Myoclonic jerks are often observed during NREM-1 sleep. Only small amounts of sleep (about 10%) are spent in NREM-1.
NREM-2 sleep is characterised by the occurrence of sleep spindles and K-complexes in the EEG. About 50% of sleep in adults is spent in NREM-2.
NREM-3 (formerly divided into NREM-3 and NREM-4) is also known as slow-wave or deep sleep. In the EEG large delta-waves dominate. Parasomnias such as night terrors and sleep-walking occur during NREM-3. About 20% of sleep is spent in NREM-3.
Respiration is assessed via abdominal and thoracic RIP belts. Together with flow signals obtained from oro-nasal sensors we can assess respiratory effort (positive readings from RIP belts). Absent airflow through nose or mouth with persisting respiratory effort (often paradoxical) is indicative of airway obstruction while absent RIP-belt signals reflect absence of respiratory effort (central apnoea).
Pulse oximetry helps assess the efficacy of the respiratory system and severity of potential impairment.
Reporting of sleep studies
All data is recorded in real-time and stored as a computer recording. Once a PSG is recorded, the recorded data has to be assessed. This involves staging of sleep and evaluation of respiratory parameters.
Respiratory events during sleep are scored according to strict criteria. Be aware that PSGs scored according to paediatric criteria can differ markedly in their assessment from those scored according to adult criteria. Obstructive episodes are assessed as hypopnoeas (>50% airflow reduction compared to previous breaths) or obstructive apnoeas (>90% reduction), with duration being two breaths.
Assessment takes place in specialised PSG-software that displays recorded data in segments called epochs (30 seconds for sleep, two minutes for respiratory parameters). And, in case you wonder: yes, sleep scientists go through all this data epoch by epoch to score sleep stage and respiratory events. This is reviewed by the sleep physician writing the report.
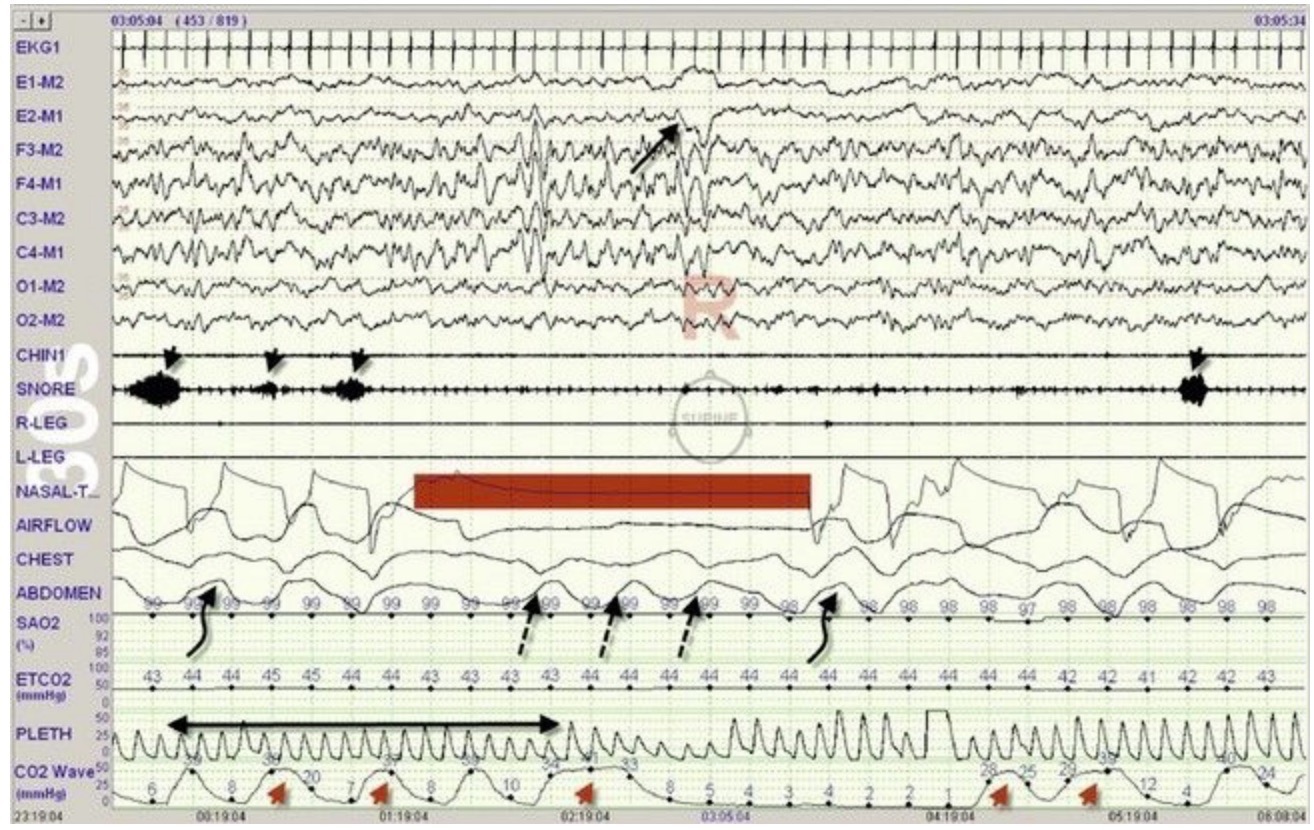
Epoch showing the occurrence of an obstructive apnoea with paradoxical abdominal/thoracic excursions (broken arrows) and loss of oro-nasal airflow (bar). Snoring (arrowheads) ceases with loss of airflow.
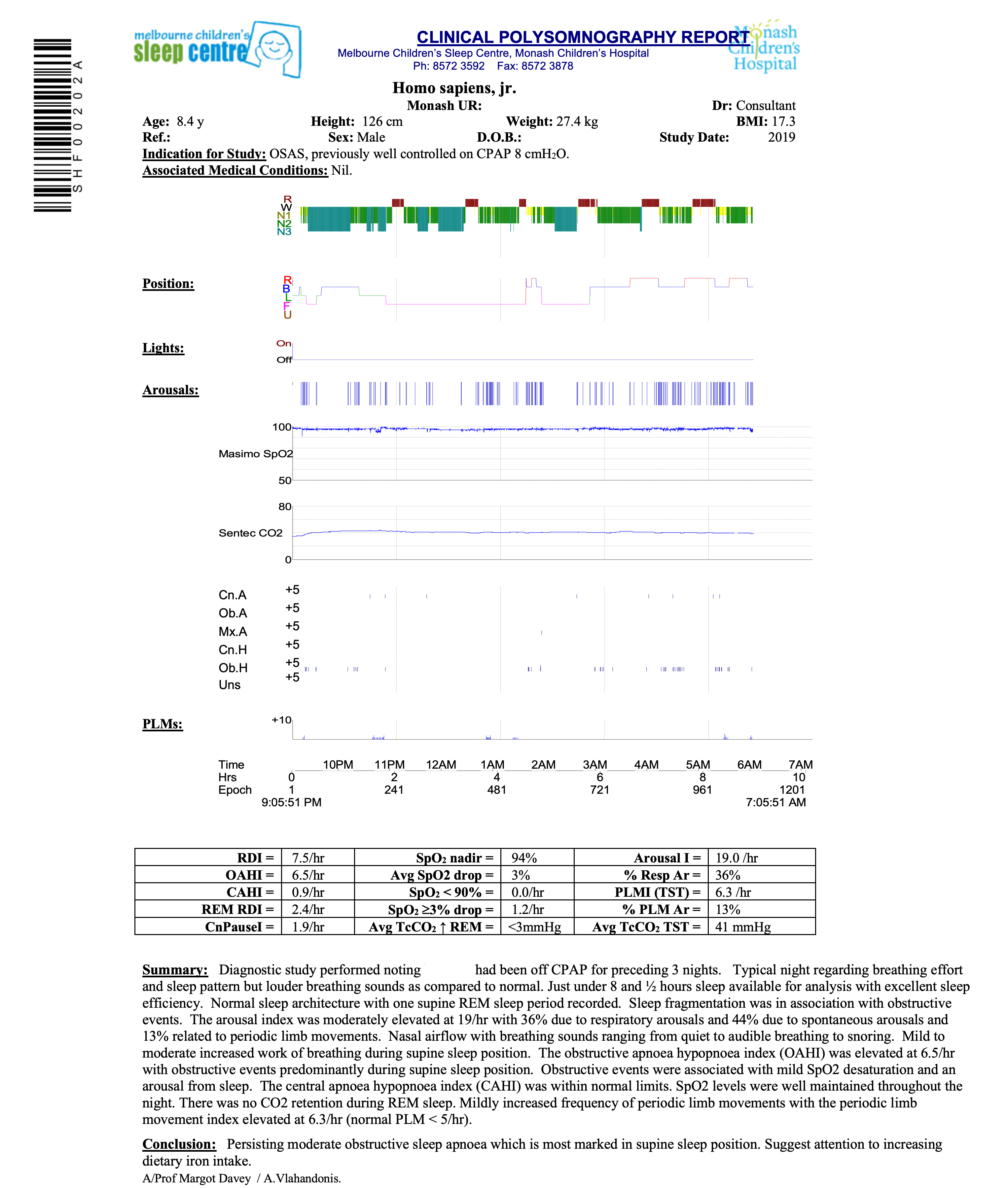
Legend
Hypnogram indicating sleep phase plotted over time on x-axis. Red areas indicate REM-sleep, grey areas periods of wakefulness, blue deep or NREM3 sleep.
Child’s body position
Arousals
Pulse oximetry. Note scale: 100% at top, 50% at bottom
Capnography
Respiratory events:
Cn.A: Central apnoea
Ob. A: Obstructive apnoea
Mx. A: Mixed apnoea
Hyp: Hypopnoea
Uns: Unsure (event does not fit criteria to be classified into any of above but is associated with desaturation or arousal)
RERA: Respiratory event related arousal (<50% reduction in airflow with associated desaturation or arousal)
Period limb movements (PLMs, >5/hr considered pathological)
Time-line (also indicates epochs)
Indices and physiological data
RDI: Respiratory Disturbance Index (all scored respiratory events Index ie. Obstructive Apnoea, Mixed Apnoea, Obstructive Hypopnoea, Central Apnoea, Central Hypopnoea, unsure)
OAHI: Obstructive Apnoea, Mixed Apnoea, Obstructive Hypopnoea Index CAHI: Central Apnoea & Central Hypopnoea Index
REM RDI: REM Respiratory Disturbance Index
CnPauseI: Central Pause Index
Avg SpO2 drop: Average SpO2 desaturation drops with scored respiratory events SpO2 <90%: SpO2 desaturation Index to <90%
SpO2 3% drop: SpO2 desaturation Index with desaturation 3%
PLMI (TST): Periodic Leg Movement Index (based on events scored in sleep)
Arousal I: Arousal Index (cortical arousals, sub-cortical activations) including those scored at end of sleep/beginning wake epoch
% Resp Ar: % Arousals following scored respiratory event
% PLM Ar: % Arousals following scored PLM
Physician’s report
Ordering a sleep study
Indications for PSG:
OSAS (obstructive sleep apnoea syndrome).
Suspected central sleep apnoea in children with developmental issues, or history of cerebral trauma or tumour.
Assessment of sleep-related respiratory reserve in children with neuromuscular disorders.
Initiation and monitoring of respiratory support including CPAP and BiPAP.
Frequent or violent parasomnias.
Periodic limb movements.
Assessment of sleep architecture in patients with EDS (excessive daytime sleepiness), usually in conjunction with an MSLT (multiple sleep latency test).
Requests for PSG from other inpatient teams
Follow this algorithm when assessing the need for PSG when requested by another inpatient team:
❖
The sleep unit is also home to Monash Children’s Hospital Non-invasive Ventilation Service. The unit provides full support to families with a child on CPAP or BiPAP. This includes fitting the appropriate mask for a child, teaching of the device and intervention to the child and family and act as a central hub where families turn to with questions around their child’s respiratory support setup.
The unit employs a variety of machines with different functionalities. Certain machines may be more appropriate for a particular child and the unit will usually advise on the best device to choose. Some of our newer models feature built in modems and can send back detailed usage reports to the unit as well as have their settings change remotely.
It may be useful to familiarise yourself with the unit’s introductory video on the use of bilevel (BiPAP) ventilators on the wards: https://www.youtube.com/watch?v=I1uiXd185eM
A variety of documents are available on Prompt, detailing the use of certain non-invasive ventilators and for decision support on questions surrounding non-invasive ventilation.
 |
Continuous Positive Airway Pressure CPAP (Paediatric) Management |
Cystic fibrosis is the most common life-limiting genetic disease in the Western world. Incidence is ≈1/2,700 births and has decreased somewhat after the introduction of antenatal screening for families at risk.
In 2013, there were just over 3,000 people with CF living in Australia. CF used to be a disease with devastating consequences to very young children. First described by American physician, Dorothy Hansine Andersen, in 1938 CF was long thought to be a disorder limited to the gastrointestinal tract. Children died in infancy from severe malnutrition secondary to exocrine pancreatic failure. Contrary to popular belief the name “cystic fibrosis” is not derived from cystic lesions forming in the lung - none of these children ever lived long enough to develop severe pulmonary sequelae of CF. It was the pancreas that showed cystic and fibrotic changes and gave the illness its name.
We have come a long way since the first description of CF by Anderson. Major milestones include the introduction of pancreatic enzyme replacement therapy, aggressive treatment of respiratory exacerbations with systemic antibiotics and physiotherapy, and inhaled therapies. More recently, the introduction of CFTR modulators have brought about a further significant improvement in CF care.
Assessing medical progress can be a bit nebulous at times, but we can very much see the impact it continues to have in improving the lives of CF patients and their families. Children with CF started to live well beyond infancy through their teenage years into adulthood. During 2013 the number of adults with CF in Australia exceeded the number of children with CF for the first time ever. Our centre now has two CF physicians taking care exclusively of adults living with CF - something virtually unheard of only 15 years ago.
CF modulators
In 2012 Ivacaftor (Kalydeco) become available to patients with at least one copy of the CFTR-mutation G551D. Ivacaftor modifies the gating of the altered CFTR-protein in affected persons, leading to increased ion-channel activity. This has marked beneficial effects on lung function, weight and pulmonary exacerbations in CF. While the total number of CF patients with this specific mutation world-wide is <5%, to many in the CF- community this drug is a “game-changer”. The first of what is now a group of CFTR-modifying drugs with a potential outlook of full correction of the disease.
Other drugs that act in a similar fashion for patients with other genotypes are actively being developed and introduced into the market, more recently the combination of Lumacaftor/Ivacaftor (Orkambi) for those homozygous for the dF508 mutation and the incoming triple combination of Elexacaftor/Ivacaftor/Tezacaftor (Trikafta) for those with a single copy of the dF508 mutation and a non-medication responsive second mutation.[^9]
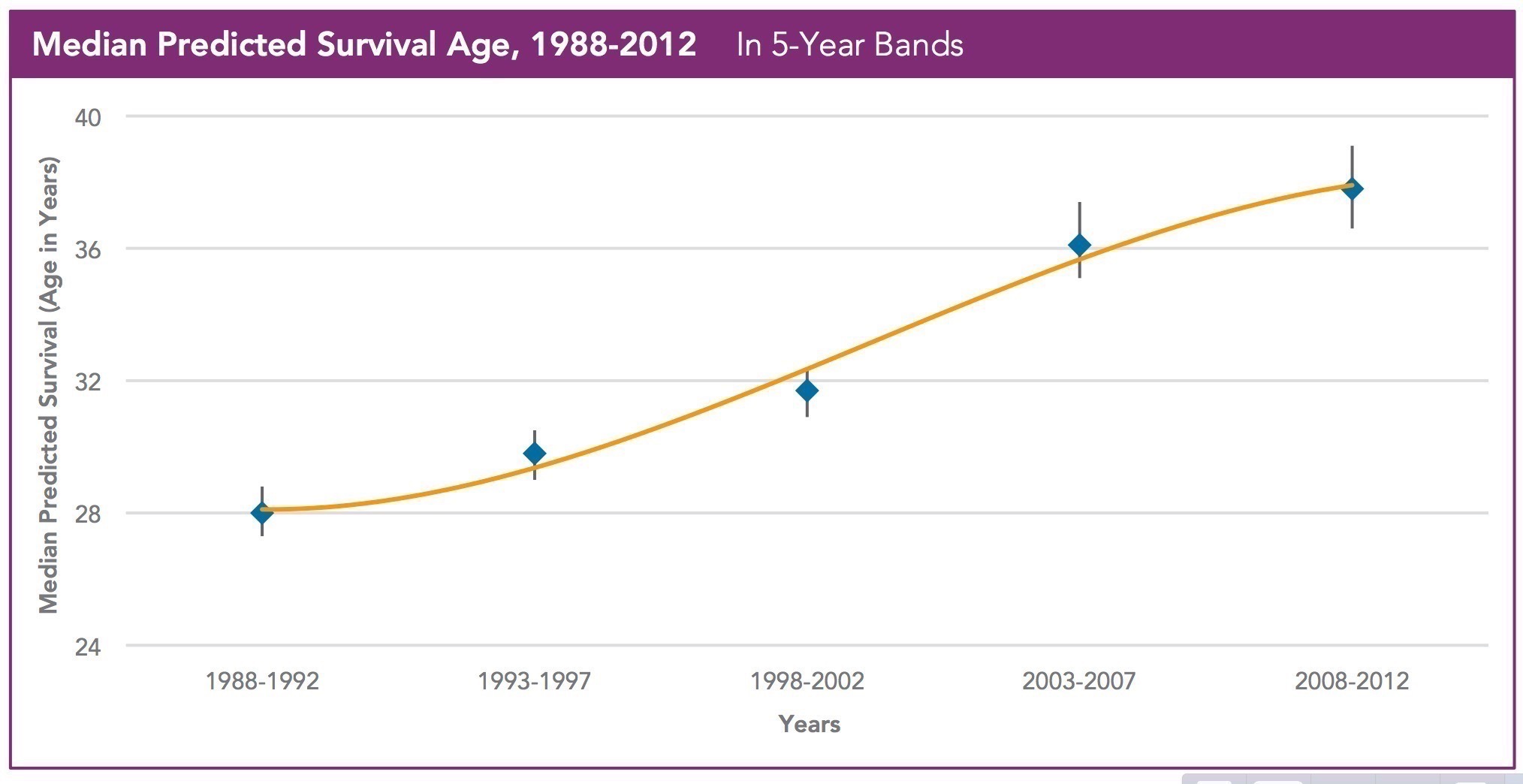
Life expectancy
Life expectancy in CF is directly related to the decade the child was born[^10], reflecting the partnership between CF-Centre, the patient and their family as well as the continuing improvements in therapies and management.
These are truly exciting times to work in cystic fibrosis.
Cystic Fibrosis is a chronic condition. Sure, you know that.
Did you also know it takes about 15 minutes for the average inhalation (“neb”) to go through a standard nebuliser? So, what’s 15 minutes? Well, that depends…
Quite a few teenagers with CF will look at the following daily regime:
Take 10-20 capsules of pancreatic enzymes
Take anywhere between 2-10 vitamin supplement tablets
Do two sets of physiotherapy, 15-30 minutes each
Inhale 4 mls of hypertonic saline BD (15 minutes each)
Inhale 2.5 mls of Dornase Alfa (10 minutes)
Inhale 5 mls of Tobramycin BD (15 minutes each)
Drink 2x 200 mls of Sustagen supplements
Spend a considerable amount of time coughing, potentially keeping you awake or waking you up overnight
We apply the term treatment burden to a daily regime like this and are done with it. Our patients have to live it every day. We see 14 days of hospital stay. For them it never stops. Try to keep that in mind when you observe the seven year old throwing a tantrum over “just a bit of inhalation therapy”. Or when you see an adolescent who decides she’s no longer going to follow any treatment advice because “it’s not making my CF go away anyway!”.
Organising your work - keeping things flowing
CF tune-ups usually happen in a fairly standardised way. The fundamentals of treatment are
Intravenous antibiotics
Physiotherapy
Additional treatments and investigations
Take a moment and think about how we are going to obtain venous access for this child.
Is there a port-a-cath? If so, speak to the child and family to find out how it is usually accessed. Families will have their own routine for most things, including port access. A particular nurse, local anaesthetic etc. might be part of the family’s preferred routine. As always, the paediatric CF co-ordinator will be an excellent first port of call.
For most line insertions you will want to discuss the most suitable line with one of our friendly anaesthetists - they’re always happy to help out with their expertise.